Carnation
Equipment needed.
1) 1 Water bottle
2) 1 White carnation
3) 3 different color of food coloring (Green, Purple, Red)
1) 1 Water bottle
2) 1 White carnation
3) 3 different color of food coloring (Green, Purple, Red)
Steps needed.
1) Pour some food coloring into the empty bottle and add some water into it.
2) Mixture the water with the food coloring.
3) Place the carnation into the bottle with food coloring mixture in it.
4) Wait patiently for the flower to turn into the color.
5) Repeat 1-4 steps for two times.
1) Pour some food coloring into the empty bottle and add some water into it.
2) Mixture the water with the food coloring.
3) Place the carnation into the bottle with food coloring mixture in it.
4) Wait patiently for the flower to turn into the color.
5) Repeat 1-4 steps for two times.
Observations.
The carnation petals turn into to green, purple, red slowly. The carnation will absorb the colored water and form the flower petal green, purple, red.
The carnation petals turn into to green, purple, red slowly. The carnation will absorb the colored water and form the flower petal green, purple, red.
Difficulties faced during the experiment.
The flower needs more time to absorb the food coloring.
The flower needs more time to absorb the food coloring.
Improvements to be made to the experiment.
Pour the food coloring more, put lesser water.
Pour the food coloring more, put lesser water.
Mind Maps
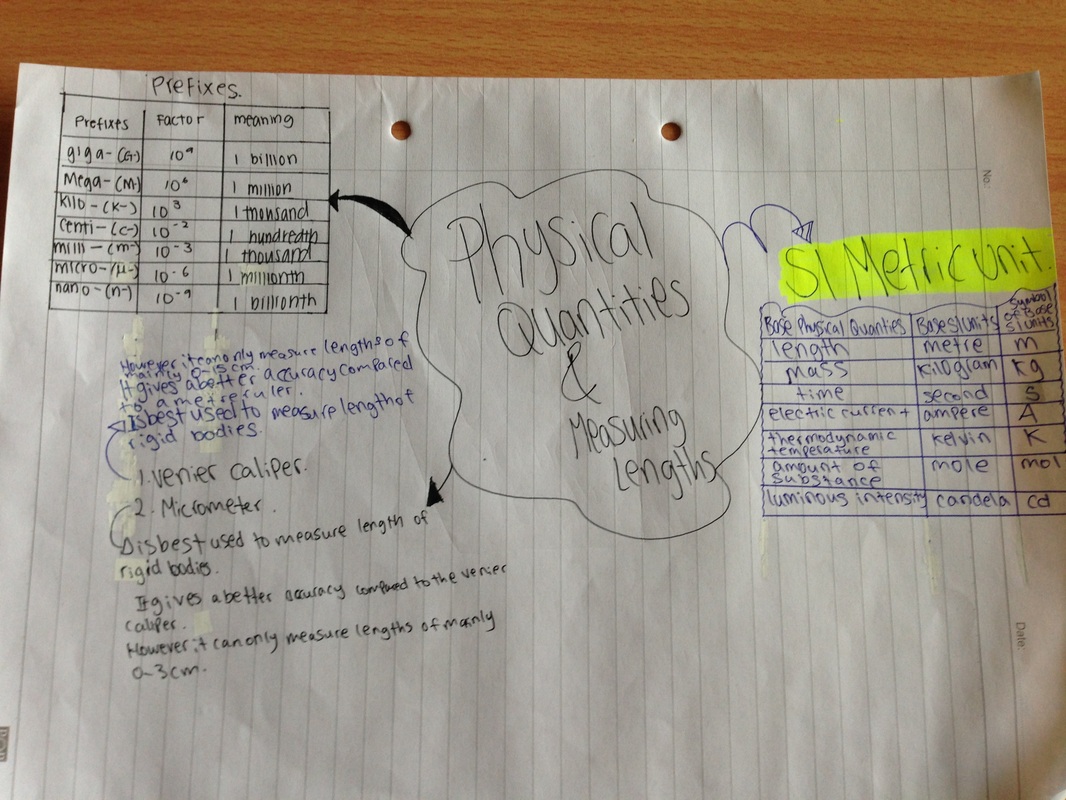
Chapter 2 Physical Quantities , Units & Measurements of Lengths
Chapter 3 Mass ,Weight & Density
Chapter 4 Simply Acids & Alkalis
Chapter 5 Separations of mixtures
Water Distillation Reflection

3 things i had learnt...
1) How does the Water Distillation functions .
2) How does the plain liquid being taken out from the coke .
3) How to make a Water Distillation figure .
2 things that i would like to find out more...
1) Whats inside the plain liquid that is being taken out ?
2) What will happen if i put a plain liquid in the flask and let it heat under the flame , does it produces different colour of liquids ?
1 thing would like to improve on my work..
1) Its not my work .
1) How does the Water Distillation functions .
2) How does the plain liquid being taken out from the coke .
3) How to make a Water Distillation figure .
2 things that i would like to find out more...
1) Whats inside the plain liquid that is being taken out ?
2) What will happen if i put a plain liquid in the flask and let it heat under the flame , does it produces different colour of liquids ?
1 thing would like to improve on my work..
1) Its not my work .
Density Tower Reflection

3 things i had learnt...
1) The method to be able to find Density is Mass/Volume .
2) Density=g/ml .
3) Different liquids have different density .
2 things that i would like to find out more...
1) Why does the liquid does not mix together ?
2) When i force the liquid to be mixed together , after a while will the liquid back to it original space ?
1 thing that i would like to improve on my work...
1) Get more layers of liquid that has different density .
1) The method to be able to find Density is Mass/Volume .
2) Density=g/ml .
3) Different liquids have different density .
2 things that i would like to find out more...
1) Why does the liquid does not mix together ?
2) When i force the liquid to be mixed together , after a while will the liquid back to it original space ?
1 thing that i would like to improve on my work...
1) Get more layers of liquid that has different density .
Acids and Alkaline Reflection

3 things that i had learnt...
1) Acids can be mix in a lot of other chemicals and produce different colours of chemicals .
2) Mixtures can be any liquids to be mix together .
3) Acid turn litmus paper RED , alkaline turns litmus paper BLUE .
2 things that i would like to find out more...
1) What does methyl orange contains ?
2) What does Litmus Paper made of ?
1 things that i would like to improve on my work...
1) Mix different amount of chemicals and see the different colours that is produced .
1) Acids can be mix in a lot of other chemicals and produce different colours of chemicals .
2) Mixtures can be any liquids to be mix together .
3) Acid turn litmus paper RED , alkaline turns litmus paper BLUE .
2 things that i would like to find out more...
1) What does methyl orange contains ?
2) What does Litmus Paper made of ?
1 things that i would like to improve on my work...
1) Mix different amount of chemicals and see the different colours that is produced .
chromatography Paper Reflection

3 things i had learnt...
1) Why did the liquid change its colour into diffrent colours .
2) Chromatography paper can allow water to rise up till the very end and it stop .
3) Chromatography paper is not an normal paper .
2 things that i would like to find out more...
1) Whats the material being used to make the Chromatography paper ?
2) Why will the water rises up through the Chromatography paper ?
1 thing that i would like to improve on my work...
1) Use a ruler with the pencil to mark the pencil marking next time .
1) Why did the liquid change its colour into diffrent colours .
2) Chromatography paper can allow water to rise up till the very end and it stop .
3) Chromatography paper is not an normal paper .
2 things that i would like to find out more...
1) Whats the material being used to make the Chromatography paper ?
2) Why will the water rises up through the Chromatography paper ?
1 thing that i would like to improve on my work...
1) Use a ruler with the pencil to mark the pencil marking next time .
Separation techniques
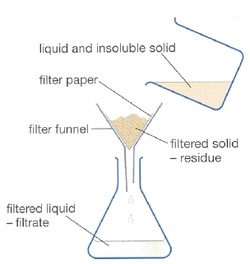
Filtration
Filtration is commonly the mechanical or physical operation which is used for the separation of solids from fluids (liquids or gases) by interposing a medium through which only the fluid can pass. The fluid that pass through is called a filtrate. [1]Oversize solids in the fluid are retained, but the separation is not complete; solids will be contaminated with some fluid and filtrate will contain fine particles (depending on the pore size and filter thickness). Filtration is also used to describe somebiological processes, especially in water treatment and sewage treatment in which undesirable constituents are removed by absorption into a biological film grown on or in the filter medium as in slow sand filtration.
Filtration is commonly the mechanical or physical operation which is used for the separation of solids from fluids (liquids or gases) by interposing a medium through which only the fluid can pass. The fluid that pass through is called a filtrate. [1]Oversize solids in the fluid are retained, but the separation is not complete; solids will be contaminated with some fluid and filtrate will contain fine particles (depending on the pore size and filter thickness). Filtration is also used to describe somebiological processes, especially in water treatment and sewage treatment in which undesirable constituents are removed by absorption into a biological film grown on or in the filter medium as in slow sand filtration.
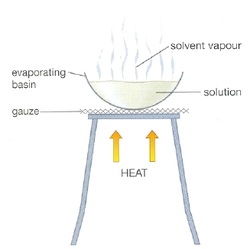
Evaporation
Evaporation is a type of vaporization of a liquid that occurs from the surface of a liquid into a gaseous phase that is not saturated with the evaporating substance. The other type of vaporization is boiling, which is characterized by bubbles of saturated vapor forming in the liquid phase. Steam produced in a boiler is another example of evaporation occurring in a saturated vapor phase. Evaporation that occurs directly from the solid phase below the melting point, as commonly observed with ice at or below freezing or moth crystals (napthalene or paradichlorobenzine), is called sublimation.
On average, a fraction of the molecules in a glass of water have enough heat energy to escape from the liquid. Water molecules from the air enter the water in the glass, but as long as the relative humidity of the air in contact is less than 100% (saturation), the net transfer of water molecules will be to the air. The water in the glass will be cooled by the evaporation until an equilibrium is reached where the air supplies the amount of heat removed by the evaporating water. In an enclosed environment the water would evaporate until the air is saturated.
With sufficient temperature, the liquid would turn into vapor quickly (see boiling point). When the molecules collide, they transfer energy to each other in varying degrees, based on how they collide. Sometimes the transfer is so one-sided for a molecule near the surface that it ends up with enough energy to 'escape'.
Evaporation is an essential part of the water cycle. The sun (solar energy) drives evaporation of water from oceans, lakes, moisture in the soil, and other sources of water. In hydrology, evaporation and transpiration (which involves evaporation within plant stomata) are collectively termed evapotranspiration. Evaporation of water occurs when the surface of the liquid is exposed, allowing molecules to escape and form water vapor; this vapor can then rise up and form clouds.
Evaporation is a type of vaporization of a liquid that occurs from the surface of a liquid into a gaseous phase that is not saturated with the evaporating substance. The other type of vaporization is boiling, which is characterized by bubbles of saturated vapor forming in the liquid phase. Steam produced in a boiler is another example of evaporation occurring in a saturated vapor phase. Evaporation that occurs directly from the solid phase below the melting point, as commonly observed with ice at or below freezing or moth crystals (napthalene or paradichlorobenzine), is called sublimation.
On average, a fraction of the molecules in a glass of water have enough heat energy to escape from the liquid. Water molecules from the air enter the water in the glass, but as long as the relative humidity of the air in contact is less than 100% (saturation), the net transfer of water molecules will be to the air. The water in the glass will be cooled by the evaporation until an equilibrium is reached where the air supplies the amount of heat removed by the evaporating water. In an enclosed environment the water would evaporate until the air is saturated.
With sufficient temperature, the liquid would turn into vapor quickly (see boiling point). When the molecules collide, they transfer energy to each other in varying degrees, based on how they collide. Sometimes the transfer is so one-sided for a molecule near the surface that it ends up with enough energy to 'escape'.
Evaporation is an essential part of the water cycle. The sun (solar energy) drives evaporation of water from oceans, lakes, moisture in the soil, and other sources of water. In hydrology, evaporation and transpiration (which involves evaporation within plant stomata) are collectively termed evapotranspiration. Evaporation of water occurs when the surface of the liquid is exposed, allowing molecules to escape and form water vapor; this vapor can then rise up and form clouds.
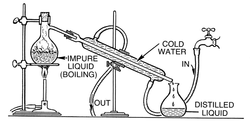
Distillation
Distillation is a method of separating mixtures based on differences in volatility of components in a boiling liquid mixture. Distillation is aunit operation, or a physical separation process, and not a chemical reaction.
Commercially, distillation has a number of applications. It is used to separate crude oil into more fractions for specific uses such astransport, power generation and heating. Water is distilled to remove impurities, such as salt from seawater. Air is distilled to separate its components—notably oxygen, nitrogen, and argon—for industrial use. Liquid chemicals for diverse uses are often distilled after synthesisto remove impurities and unreacted starting materials. Distillation of fermented solutions has been used since ancient times to producedistilled beverages with a higher alcohol content. The premises where distillation is carried out, especially distillation of alcohol, are known as a distillery. A still is the apparatus used for distillation.
Distillation is a method of separating mixtures based on differences in volatility of components in a boiling liquid mixture. Distillation is aunit operation, or a physical separation process, and not a chemical reaction.
Commercially, distillation has a number of applications. It is used to separate crude oil into more fractions for specific uses such astransport, power generation and heating. Water is distilled to remove impurities, such as salt from seawater. Air is distilled to separate its components—notably oxygen, nitrogen, and argon—for industrial use. Liquid chemicals for diverse uses are often distilled after synthesisto remove impurities and unreacted starting materials. Distillation of fermented solutions has been used since ancient times to producedistilled beverages with a higher alcohol content. The premises where distillation is carried out, especially distillation of alcohol, are known as a distillery. A still is the apparatus used for distillation.
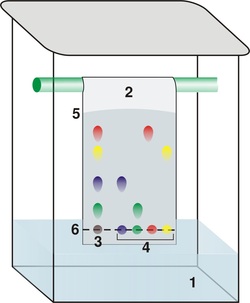
Paper ChromatographyPaper chromatography is an analytical method technique for separating and identifying mixtures that are or can be coloured, especially pigments. This can also be used in secondary or primary colours in ink experiments. This method has been largely replaced by thin layer chromatography, but is still a powerful teaching tool. Double-way paper chromatography, also called two-dimensional chromatography, involves using two solvents and rotating the paper 90° in between. This is useful for separating complex mixtures of compounds having similar polarity, for example, amino acids. If a filter paper is used, it should be of a high quality paper. The mobile phase is developing solutions that can travel up to the stationary phase carrying the sample alongside with it.
Solutions and Suspensions
What are solutions?
Ans: Solutions are mixtures in which soluble particles are completely dissolved in a liquid or gas.
What is the name given to the substance which dissolves in the solvent?
Ans: Solute
What is the name given to the substance which dissolves the solute?
Ans: Solvent
List out 3 characteristics of a solution.
1) Homogeneous (uniformly mixed - only see one layer)
2) No residue after filtration(cannot be separated by a filter)
3) No separation of solute and solvent when left to stand(always maintain a uniform appearance)
Salt is mixed with water to form a salty solution.
i) Which is solute?
Ans : Salt
ii) Which is solvent?
Ans : Salty solution
What are suspensions?
Ans : Suspension are mixtures in which insoluble particles are suspended in a liquid or gas.
List out the 3 characteristics of a suspension .
1) Non-homogeneous(not uniformly mixed)
2) Residue present after filtration(component can be easily separated)
3) Separation of solute and solvent when left to stand(more than one layer appear after being made to stand for awhile)
Some flour is mixed with oil. After the mixture has a chalky appearance and eventually the flour settles at the bottom of the oil.
i) Which is the insoluble substances?
Ans : Flour
ii) Which is the solvent?
Ans : Oil
List 2 differences between a suspension and solution?
1) Suspension is non-homogeneous Solution is homogeneous
2) Suspension residue present after filtration Solution no residue after filtation
Ans: Solutions are mixtures in which soluble particles are completely dissolved in a liquid or gas.
What is the name given to the substance which dissolves in the solvent?
Ans: Solute
What is the name given to the substance which dissolves the solute?
Ans: Solvent
List out 3 characteristics of a solution.
1) Homogeneous (uniformly mixed - only see one layer)
2) No residue after filtration(cannot be separated by a filter)
3) No separation of solute and solvent when left to stand(always maintain a uniform appearance)
Salt is mixed with water to form a salty solution.
i) Which is solute?
Ans : Salt
ii) Which is solvent?
Ans : Salty solution
What are suspensions?
Ans : Suspension are mixtures in which insoluble particles are suspended in a liquid or gas.
List out the 3 characteristics of a suspension .
1) Non-homogeneous(not uniformly mixed)
2) Residue present after filtration(component can be easily separated)
3) Separation of solute and solvent when left to stand(more than one layer appear after being made to stand for awhile)
Some flour is mixed with oil. After the mixture has a chalky appearance and eventually the flour settles at the bottom of the oil.
i) Which is the insoluble substances?
Ans : Flour
ii) Which is the solvent?
Ans : Oil
List 2 differences between a suspension and solution?
1) Suspension is non-homogeneous Solution is homogeneous
2) Suspension residue present after filtration Solution no residue after filtation
Density.
I had learned to be patience when we are trying to see whether what kind of liquid is denser while the other is not.
The method to be able to find Density is Mass/Volume.
Density=g/ml
When we are doing the density tower,we have to be very careful when we pouring the liquid into the test tube.
If we pour it too quickly,the liquid might sink to the bottom.
And teamwork is very important in doing this experiment.
The method to be able to find Density is Mass/Volume.
Density=g/ml
When we are doing the density tower,we have to be very careful when we pouring the liquid into the test tube.
If we pour it too quickly,the liquid might sink to the bottom.
And teamwork is very important in doing this experiment.